Importers
-
REACH
- Understanding REACH
- Substance Identification
- Registration
- Evaluation
- Authorisation
- Restriction
- Communication in the supply chain
- Candidate List substances in articles
- Legislation
- Alternatives to animal testing under REACH
- Enforcement
- Nanomaterials
- Assessment of regulatory needs
- PBT assessment
- Endocrine disruptor assessment
- Cooperation with authorities and stakeholders
- Addressing substances of concern
Importers
Importers
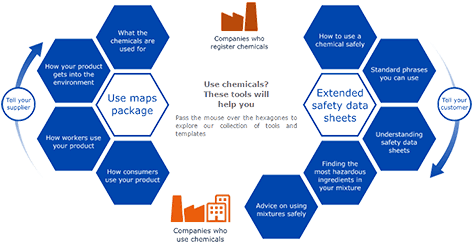
Through the Chemical safety report/Exposure scenario roadmap (CSR/ES) Roadmap stakeholders have produced a number of tools to support supply chain communication.
For an importer, where substances are concerned, these include the same tools as for a manufacturer:
- sector use maps – for upstream communication
- the ESCom standard phrase catalogue and ESCom XML standard package
- illustrative examples of chemical safety reports (CSRs) and exposure scenarios (ESs)
- ES templates
- the table of contents for the Annex to a SDS (ES restructured short titles SST)
- the Interactive guide on Safety data sheets and Exposure scenarios
For mixtures, additional tools include:
- the information on mixture classification
- the Lead Component Identification (LCI) methodology
- the Safe Use of Mixture Information under REACH approach and template
For articles, there are no tools developed by the CSR/ES Roadmap. The most appropriate way to communicate can vary, depending on the content and the addressee of the information. More information can be found in ECHA Guidance on requirements for substances in articles, section 3.4.1.
For more information, see the links provided.
See also the ECHA Guidance pages for more support on how to fulfil your obligations under the relevant legislation.
Related
- Use maps
- ESCom standard phrase catalogue (CEFIC)
- Examples of chemical safety reports
- Examples of exposure scenarios
- Formats and templates
- Guidance on REACH
- Mixture classification
- DUCC Publications
- Lead Component Identification (LCI) methodology (CEFIC) [PDF]
- Guidance on requirements for substances in articles [PDF]